Acoels
Introduction
This module is composed of four quite distinct chapters. In Chapters 1 and 2, you will be introduced to the phenomenon of marine photosymbioses, and then to our star Acoela model S. roscoffensis, by Xavier. Then, Simon will take the reins in the following Chapters 3 and 4 to give you an overview of Acoela phylogeny, as well as briefly compare different acoel experimental models and the molecular techniques available.
Chapter 2: An introduction to S. roscoffensis
Chapter 3: Acoela phylogeny
 Hi, my name is Simon Sprecher, and I am a Professor in Biology and Neurogenetics at the University of Fribourg in Switzerland. A key question that has been fascinated me for years is how the brain – or more generally how nervous systems – evolved. Due to their phylogenetic position, acoel models are of great importance in studying brain evolution.
Hi, my name is Simon Sprecher, and I am a Professor in Biology and Neurogenetics at the University of Fribourg in Switzerland. A key question that has been fascinated me for years is how the brain – or more generally how nervous systems – evolved. Due to their phylogenetic position, acoel models are of great importance in studying brain evolution.
You’ve already had an introduction to one of the members of Acoela, S. roscoffensis. More generally, acoels are small, mostly marine worms that can be found in many ecological niches, and at first sight can appear quite similar to Platyhelminths, but since the first molecular phylogenies at the beginning of this century it became clear that they are a separate clade.
Acoela belong to the largely enigmatic phylum Xenacoelomorpha, which consists of three clades: Xenoturbellida, Nemetodermatida and Acoela. The phylogenetic position of these clades has been highly debated, a discussion that is still not completely resolved.
Among these three clades, Acoela has about 400 described species and is therefore the most species-rich of the three clades. Nemetodermatida is comprised of a few dozen species and for a long time Xenoturbellida consisted of a single described species (there have since been a few more deep-sea species discovered).
So, what makes these marine worms so special? Above all, the fact that they are the only bilaterian clade that lacks specialized excretory systems, such as “nephridia”, simpler forms of kidneys.
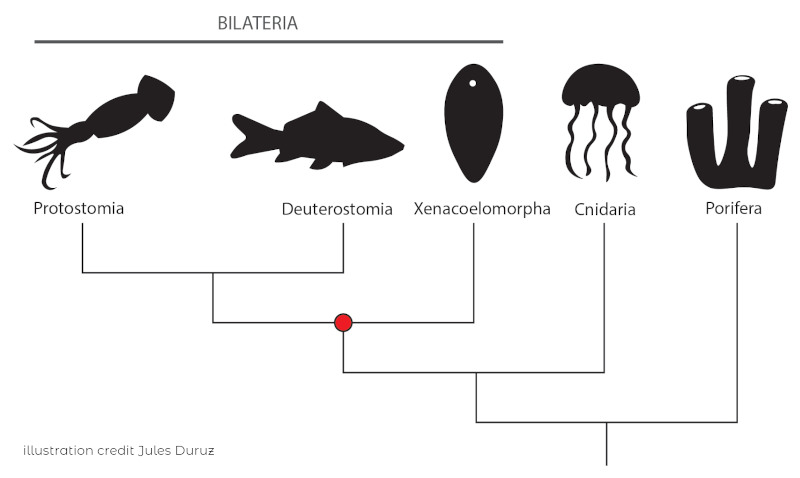
Thus, in one phylogenetic scenario, they are the sister group to all “nephrozoa”, and thus would represent the clade that split off prior to the diversification of all other bilaterians. As mentioned, the precise phylogenetic position is still debated.
One often easily visible, external feature that all Xenacoelomorpha share is the presence of a statocyst in its head.
Chapter4: Acoel models and molecular techniques
There are currently two Acoela species that we use in the laboratory as models, or experimental systems.

These species are Symsagittifera roscoffensis, which you already know, and Isodiametra pulchra. As you’ve seen in other modules in this course, all species have some relative advantages as models and thus can be studied to answer specific questions. Symsagittifera roscoffensis has the advantage that it can be collected in large amounts throughout the year and that it has specialized to live in symbiosis with single-celled algae. It can be kept in the lab easily for several months. Isodiametra pulchra is a bit smaller and can be grown simply on petri-dishes covered a thin layer of single-celled algae, which it needs to feed on. As long as the algae do well typically the animals thrive. It has a generation time of about 5 weeks and can be kept over several generations in the lab.
Both species are used as models for regeneration studies. There are some interesting key differences between these two models: Symsagittifera regenerates head and tail well, while Isodiametra appears not to be able to regenerate its head, but rapidly regenerates its posterior part. These differences also allow us to address how similar or conserved the mechanisms of regeneration is.
Did you know that Symsagittifera can regenerate its head and brain within a few weeks? Using behavioral experiments, we have shown that within 3 weeks the brain is already functioning - the animals once again show their typical phototaxis behavior, which means that both the eyes and the brain are regenerated and functional!
Let’s look at some of the molecular tools available. In view of molecular and genetic techniques, currently the biggest advances are made in sequencing transcriptomes and genomes. For both species there are very good transcriptomes published and available for the scientific community, as well as protocols for immunostainings and in situ hybridization that are available; together with transcriptomic data, this allows us researchers to examine which genes are expressed during which stages and their expression patterns.
It is important to note that both species can also be cultured in the lab in quite large quantities and the life cycle has been completed. As you know, this is an important prerequisite to study embryogenesis and also allows us to inject oocytes, which is a the most critical step for doing transgenics or using CRISPR and morpholinos.
A more recent technique which we used is single-cell transcriptomics; this technique allows us to identify all genes which are expressed in any cell type of the body. We used this technique on the Isodiametra hatchling, which consists of only about 1000 cells. We sequenced roughly 15’000 cells and achieved therefore a 14x coverage for each cell. Using this approach, we could describe 10 major cell types and the genes they express with great precision. What we felt was particularly interesting, was a surprising diversity of neurons. Functional genetic studies remain currently still a bit challenging. For Isodiametra pulchra RNAi protocols have been published. By using RNAi, several genes required for regeneration were studied.
As is the case for many emerging model organisms, molecular tools remain one of the biggest challenges. It is clear that CRISPR/Cas9 mediated genome editing is one of the most important next technical steps to be achieved.
Teachers: Xavier Bailly and Simon Sprecher
